The Devil is in the Details: Pfizer's Fraudulent EUA of its BNT162B2 Vaccine for Babies
Pfizer has removed our ability to check long-term safety of any of its vaccines!
Pfizer submitted a request to the FDA on May 27, 2022, to modify the Emergency Use Authorization (EUA) for the Pfizer-BioNTech COVID-19 Vaccine (BNT162b2) for the prevention of coronavirus disease 2019 (COVID-19), which is caused by coronavirus-2 and causes severe acute respiratory syndrome (SARS-CoV-2). The modification would allow BNT162b2 to be used in babies and children aged 6 months to 4 years in a three-dose primary series (3 milligrams per dose). Pfizer presented safety, clinical outcomes, and preliminary descriptive efficacy data from the C4591007 randomized, double-blind, placebo-controlled trial in response to the FDA's request. Participants aged 6 months to 4 years were given two doses of BNT162b2 (3 milligrams mRNA per dosage) or a saline placebo three weeks apart. The protocol was revised to add a third primary series dose administered at least 8 weeks after Dose 2 to individuals aged 6 months to 4 years after an analysis of the post-Dose 2 safety and efficacy data.
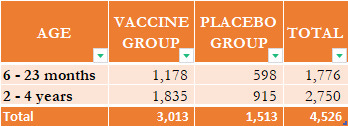
CORRECTION: I originally stated that 3,000 children had dropped out of the trial, but this was due to a misinterpretation of the data tables on Page 39 of the FDA's briefing presentation. The trials had two major groups of participants, which was the source of my confusion. These were the groupings:
The EUA request comprised phase 2/3 safety data from 1,178 BNT162b2 patients and 598 placebo patients aged 6 to 23 months (hereafter 6-23 months) and 1,835 BNT162b2 patients and 915 placebo patients aged 2 to 4 years (hence 2-4 years) who received at least one dose of the investigational product.
Pfizer presented information the the US Food and Drug Administration (FDA) that its BNT162B2 Covid-19 vaccine is effective in children aged six months to four years. Pfizer's study data that was presented to the FDA is, predictably, quite concerning. Pfizer recruited 4,526 children ranging in age from six months to four years old for its Phase 1/2/3 Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate Against COVID-19 in Healthy Children and Young Adults. Search for NCT04816643 on ClinicalTrials.gov for further details. The most concerning statistic is that 3000 of these children, or 66.28 percent, did not complete the trial.
There was no explanation for why two-thirds of the study participants dropped out, and regardless of the reasons, you'd expect the trial would be declared null and void. We must demand that Pfizer explain why 3,000 children withdrew from the trial, as this is crucial information that the public has a right to know. So, what was the outcome of the trial? Children with a slightly higher heart rate or few more breaths per minute were classified as having severe Covid. A total of eight (8) children were diagnosed with “severe covid” during the trail. Six (6) of the children were from the vaccine group while one two children were from the placebo group. Therefore, the chances of this vaccine producing severe Covid are higher than the chances of it not. Furthermore, one vaccinated child was hospitalized with sever Covid after coming down with high fever and suffering a seizure.
This information can be found on page 6 of the document in paragraph 2.
NOTE: To verify every claim that I have made in this article, please, refer to Vaccines and Related Biological Products Advisory Committee Meeting June 15, 2022 FDA Briefing Document EUA amendment request for Pfizer-BioNTech COVID-19 Vaccine for use in children 6 months through 4 years of age, published by the United States Food and Drug Administration.
So now we'll look at what Pfizer meant by "any Covid." Pfizer completely distorted the data. They inoculated the children and then waited three weeks after the first dose before administering the second dose.
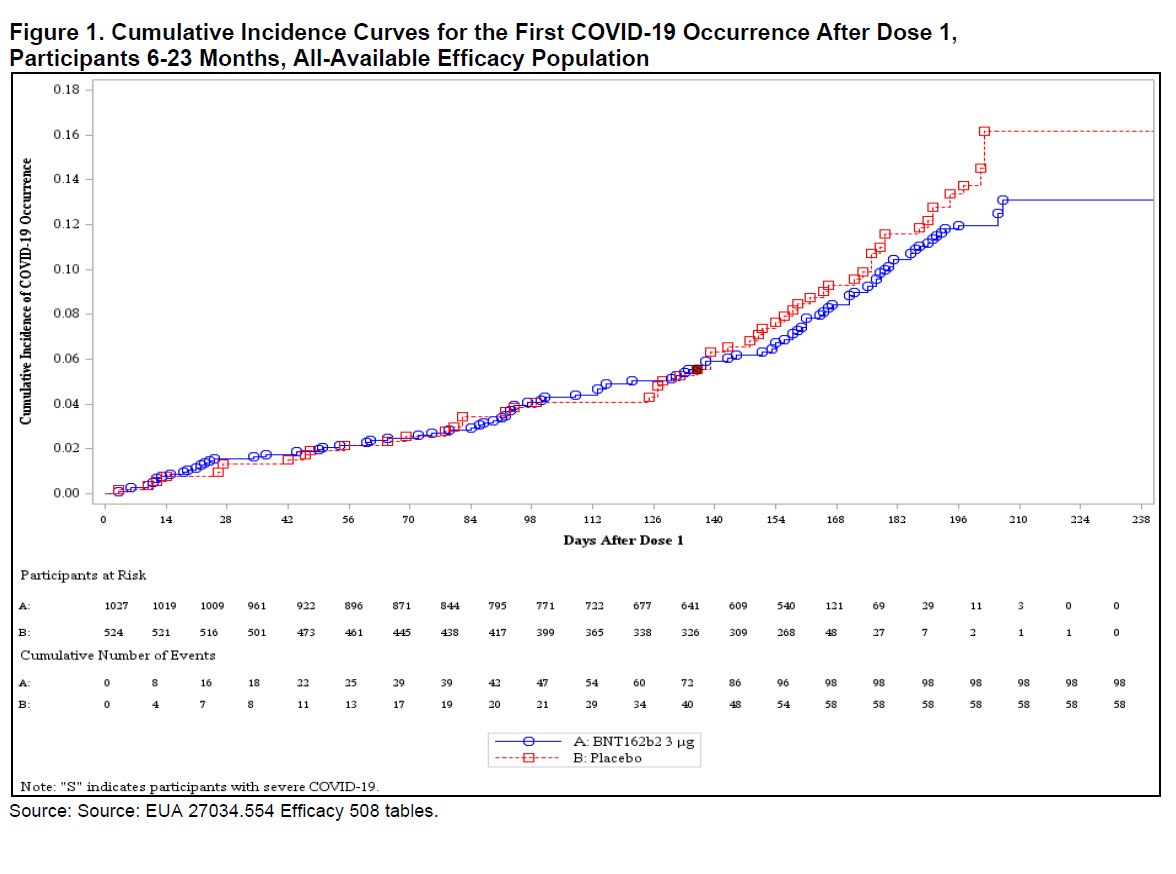
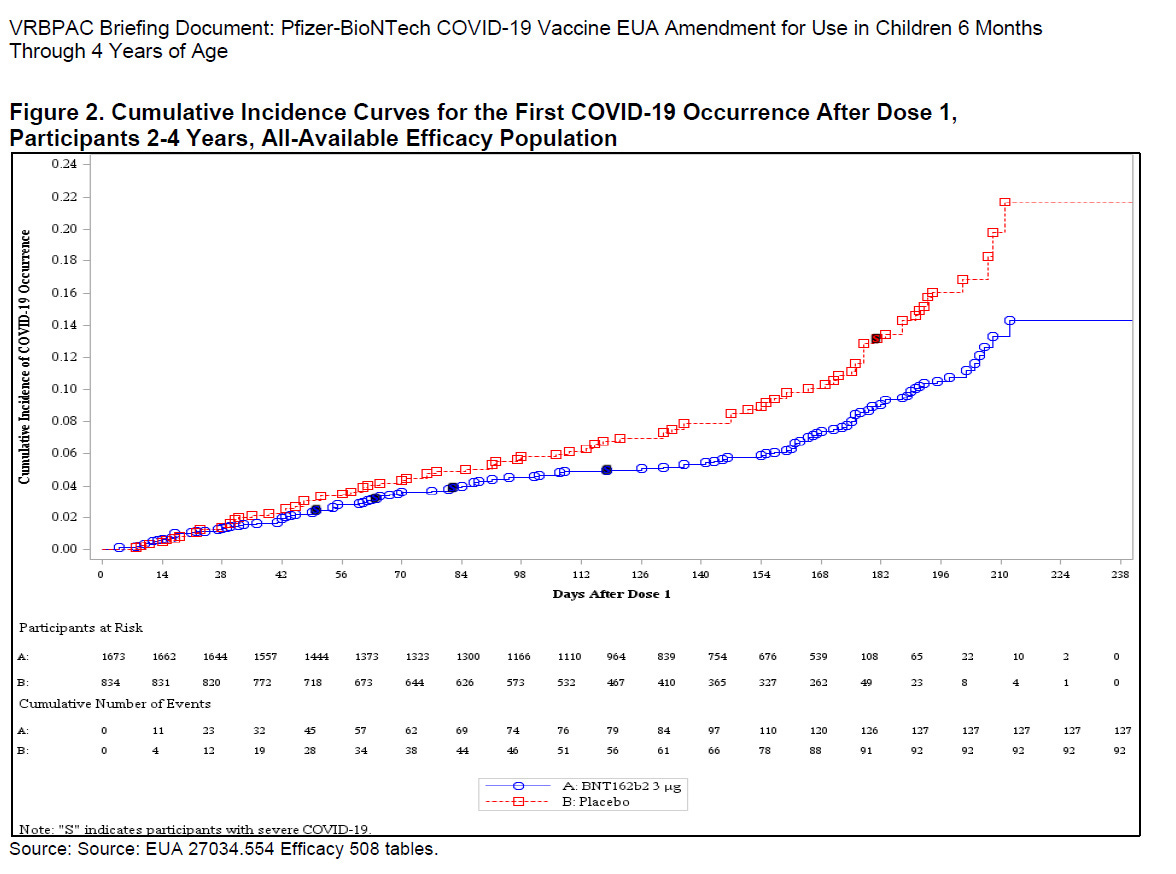
The most shocking information is that hundreds of children contracted Covid during the trial and Pfizer totally ignored that information when it was determining the efficacy of the vaccine. Between Day zero on Day 28, 39 vaccinated children contracted Covid-19 compared to 19 children in the placebo group, after the first dose, which means that the vaccine had no effect on preventing infection. But unsurprisingly, Pfizer and the FDA ignored the data. Furthermore, there was an eight-week gap between the second and third doses, during which time many of the children in the vaccine arm of the trial became infected with Covid. Pfizer also ignored that data. They also ignored the Covid infections that occurred several weeks after the third dose, meaning that they had overlooked 97 percent of the Covid infections that occurred during the experiment and only looked at a small percentage of the data that would provide good efficacy results.
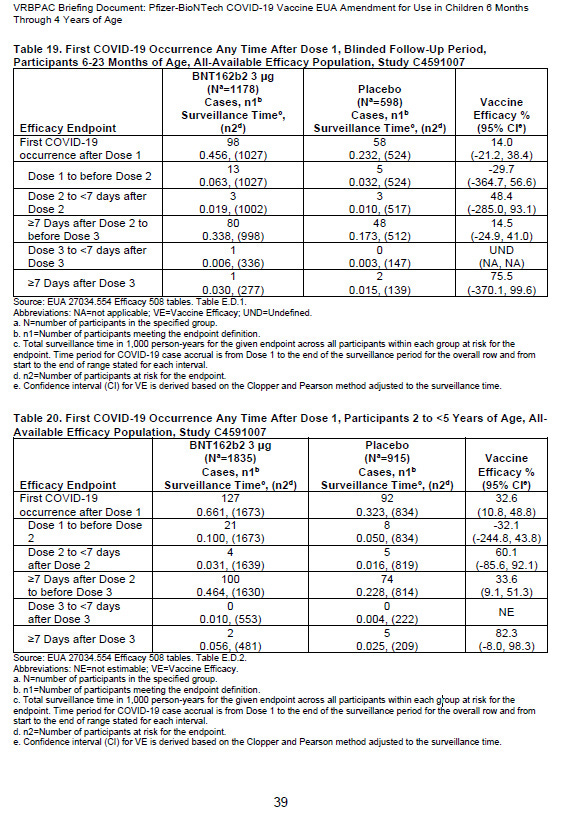
Pfizer compared only three children in the vaccine arm of the trial , who contracted Covid, against seven in the placebo arm and because they chose such a small dataset, the results of the study lead to a conclusion of 75% vaccine efficacy. However, over the entire course of the trial, a total of 225 vaccinated children contracted Covid-19 compared to 150 in the placebo group.
This would mean that Pfizer’s vaccine had a efficacy of :
Vaccine 225/3013 = 0.746
Placebo 150/1513 = 0.991
Therefore 0.746/0.991 = 0.75, efficacy equals 1 - 0.75 = 25%
The vaccine has no benefit to children. Furthermore, during the two-month follow-up period, Pfizer also kept track of how many of these kids caught COVID twice. Twelve children tested positive for COVID twice, and all but one (11/12) had been vaccinated, with the majority receiving three doses of the BNT162B2 vaccine. Therefore, it begs the question: how could Pfizer claim that the vaccine was effective against Covid when there were twelve children who have had COVID twice, with eleven of them having received vaccinations?
Let's go over everything one more time. They enrolled 4,526 children in the trial, but 3000 dropped out for reasons that were never revealed. Pfizer then claimed that its vaccine was effective, by presenting data to the FDA that showed that three children in the vaccine arm of the trial contracted Covid compared to seven in the placebo group, a four-child difference. Keep in mind that none of this takes into account the fact that Covid-19 has negligible impact on children, and a study published in the New England Journal of Medicine showed a 82% spontaneous abortion rate among vaccinated pregnant women. How could an "ethics" committee accept this type of research on babies given the lack of long-term safety evidence? What’s worst, Pfizer used the results of this trial as evidence in its application to the FDA for an emergency use authorization of its BNT162B2 vaccine for children under five years of age. However, there is no COVID “emergency” for children and an Emergency Use Authorization (EUA) should only be granted when there is a high risk of serious injury or death. The data do not support an EUA of Covid-19 vaccines for children.
In fact, Pfizer had to devise new methods of quantifying the problem (antibody tithers) because there was no major injury or death in the vaccine trial. It's worth remembering that the vaccines were once touted as having the ability to reduce transmission. However, the data categorically show that the vaccines do not reduce transmission, and even the World Health Organization (WHO) has withdrawn its assertion that these mRNA biologics help to reduce the transmission of the SARS-CoV-2 virus. So, Pfizer’s claim of a reduction in transmission arising from its trial on babies is fraudulent and misleading. Moreover, the argument for vaccinating babies was to provide a shield for older, more vulnerable adults, but everyone can agree that it is clearly completely wrong to use young children as human shields. And since we all know, from the data, that the vaccines do not prevent infection or transmission, why on earth would the FDA approve them for babies?
In terms of safety, the children were observed for six weeks before being unblinded and vaccinated. Unblinding, in a clinical trial, means that the participants in the placebo arm of the trial - the group that will not be getting the real vaccine - were told that they did not receive the actual covid vaccine but had been given a placebo ( an inactive substance or other intervention that has no therapeutic effect but was given in the same manner as the real drug). What Pfizer did was to track the children in the placebo group for an average of six weeks, after which it told then that they didn’t get the real vaccine and proceeded to vaccinate those children. This action on the part of Pfizer effectively eliminated the safety control group forever. The practice of removing the placebo group at some point during the clinical trail has been done in every covid-19 vaccine trial to date. This is a criminal act by Pfizer and all the other vaccine manufacturers which has been ratified by the WHO, FDA, EMA, UHSA, and CDC.
We cannot determine the long-term safety of covid-19 mRNA biologics because all trials have unblinded and vaccinated their participants. Most people are unaware that none of the Covid-19 trials have been completed. I believe the first will end in 2023. This latest trial on babies by Pfizer is set to end in 2024. Therefore, everyone who has received a Covid-19 vaccine is taking part in an active experiment, with no guarantee of long-term safety data. Worse, they (health officials) sought to vaccinate every single person on the planet. This effectively gives them a clean canvas on which to conjure up various scientific arguments to "explain away" the injuries and deaths caused by mRNA gene-therapies (fake vaccines). This can be seen in real time, as they use the name Sudden Adult Death Syndrome (SADS) to "explain" why otherwise healthy young people are "suddenly" dying. When their bioweapon vaccines kill our children, what excuse will they give us?